Publications
- All
-
Following
You must log in
before getting
tailored content. -
Interests
You must log in
before getting
tailored content. - Most liked
- Most viewed
Directory:
Tags:
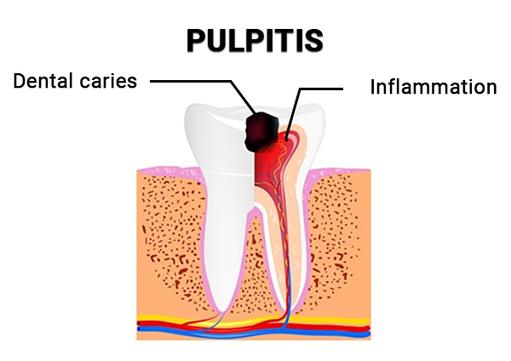
When to Seek Urgent Dental Care
It is vital to understand what constitutes a dental emergency so that you can be prepared and respond appropriately. While a chipped, cracked, or broken tooth may seem to be a life-threatening issue, you can usually make an appointment with your Stamford-based dentists Ali Modiri, DDS, and Marjan Moghadam, DDS within the following day or two to address it.
The most prevalent cause for seeking emergency dental care is toothache relief. Tooth pain can be caused by a tooth infection or tooth decay. These causes build gradually, implying that you haven’t been keeping up with your regular checkups and cleanings, and a simple problem has turned into a painful experience that requires immediate attention. Your Stamford dentist can still help you.
Types of Dental Emergencies
Other emergency problems that require immediate dentist assistance, in addition to tooth decay or infection, include:
Trauma. Accidents such as a car crash or a fall (whether down the stairs or off a bike) can result in serious facial and dental injuries. It is possible to have a broken tooth or a jarring misalignment of teeth. As a result of such a trauma, you may see blood in your mouth and experience jaw pain.
Sports-related injuries. An injury sustained while participating in sports is another common reason for urgent dental care. A pitch can hit you in the face, or an errant elbow can strike you. The damage can be serious, such as fracturing a tooth, loosening a dental crown, or knocking out your dental bridge completely.
Knocked-out teeth. While your teeth are strongly anchored in your gums, a direct hit can completely knock permanent teeth out of their sockets. To save the teeth, you must act quickly. Pick them up with caution, being careful not to damage the root or remove any tissue membrane. Clean the teeth gently with water and reinsert them in your mouth. If this isn’t possible, soak the knocked-out teeth in milk to keep them from drying out. Your Stamford dentist may still be able to restore them.
Pulpitis. Pulpitis is a condition that can result in heart conditions, meningitis, and even brain abscesses. It occurs when the pulp becomes inflamed as a result of untreated dental caries, resulting in a severe toothache and abscess. Because pulpitis is a serious condition, seek treatment at a nearby dental emergency clinic as soon as possible. You may also need gum disease treatment to resolve your periodontitis and avoid further infection.
Bleeding and swelling. Your body may react negatively to any oral surgery, including the procedure to get a dental implant. You may notice more blood in your mouth than expected, as well as significant swelling. These problems can be solved with urgent dental care.
Your treatment depends on the cause of the emergency and how severe your pain is. Stamford Dental Arts uses cutting-edge equipment to provide treatments such as emergency tooth extraction, root canal procedure, or cavity filling. This Stamford emergency dental center has everything you need.
Stamford Dental Arts
44 Strawberry Hill Ave, Suite 1,
Stamford, CT 06902
(203) 504–8745
Web Address:
https://www.stamforddentalarts.com/
Our location on the map:
https://goo.gl/maps/Y2YfWteR7rjhUzUT6
https://plus.codes/87H83F68+PM Stamford
Nearby Locations:
Ridgeway | Strawberry Hill | Glenbrook | East Side Of Stamford | Downtown
06905 | 06902 | 06906 | 06901, 06905
Working Hours:
Monday: 2:00 pm — 8:00 pm
Tuesday: 8:00 am — 4:00 pm
Wednesday: 8:00 am — 4:00 pm
Thursday: 8:00 am — 8:00 pm
Friday: 8:00 am — 4:00 pm
Saturday: CLOSED
Sunday: CLOSED
Payment: cash, check, credit cards.
Directory:
Tags:
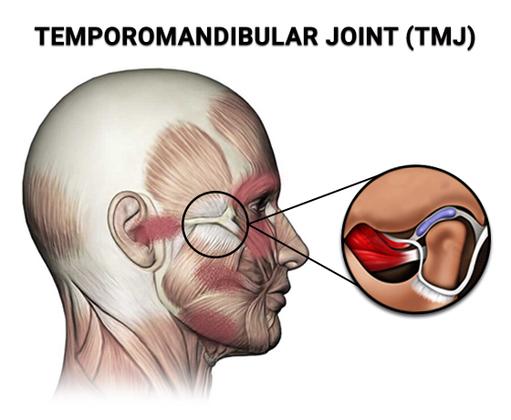
Treatment for TMJ dysfunction may be required when you experience symptoms severely enough. If you ever feel extreme pain in the joints and muscles on either side of your jaw, call on your dentist as soon as you can.
Seeking Treatment for TMJ
Pinpointing the origin of your pain can be difficult, as there are often several reasons for a case of TMJ dysfunction to act up. While the condition can affect anyone, it surfaces more frequently in women than in men. The latest research indicates that certain hormonal fluctuations in women can create functional problems with the temporomandibular joint.
Our dental center is well equipped and expertly staffed to correctly diagnose TMJ dysfunction and prescribe TMJ treatment. Whatever the reason for your TMJ pain, the experienced dentist takes effective action to diminish your pain and address the source. Don’t hesitate to visit your family dentist, since jaw pain can spread to your face, your ears, and even your eyes.
Causes of TMJ Dysfunction
TMJ pain can come from a specific incident or from long-term wear and tear. For younger adults, TMJ is usually due to overuse and stress on the jaw muscle because of teeth grinding or jaw clenching. Sleep apnea sufferers also can experience TMJ for the same reason, resulting in muscle spasms in the jaw area.
For older people, the condition is usually linked to the breakdown of the joint muscle and cartilage due to rheumatoid arthritis or osteoarthritis. Both of these diseases can weaken the jawbone, making you susceptible to fractures at the slightest trauma or accident. You can also suffer from jaw pain if your dentures or denture implants don’t fit you properly. Other causes for TMJ include:
Sports-related injuries where your jaw was directly hit
Injuries you get from accidents such as a car crash or bad fall
A misalignment of teeth, causing stress to your jaw whenever you chew
Tooth decay or a tooth infection that results in pulpitis or periodontitis
Stamford Dental Arts
44 Strawberry Hill Ave, Suite 1,
Stamford, CT 06902
(203) 504–8745
Web Address:
https://www.stamforddentalarts.com/
Our location on the map:
https://goo.gl/maps/Y2YfWteR7rjhUzUT6
https://plus.codes/87H83F68+PM Stamford
Nearby Locations:
Ridgeway | Strawberry Hill | Glenbrook | East Side Of Stamford | Downtown
06905 | 06902 | 06906 | 06901, 06905
Working Hours:
Monday: 2:00 pm — 8:00 pm
Tuesday: 8:00 am — 4:00 pm
Wednesday: 8:00 am — 4:00 pm
Thursday: 8:00 am — 8:00 pm
Friday: 8:00 am — 4:00 pm
Saturday: CLOSED
Sunday: CLOSED
Payment: cash, check, credit cards.
Directory:
Tags:
The clinic uses the latest technologies in order to ensure your experience a pleasant, effective and efficient treatment. An experienced team of dentists of Stamford Dental Arts has more than enough competencies needed to address any dental issue you or your family members might have. The team of highly qualified experts devotes thousands of hours in order to keep abreast of the latest technologies in the dental industry, but their main focus is still on customers. This top-rated dental clinic has all the latest technologies in place for an accurate diagnosis and can offer the most applicable procedures based on the best practices. Additionally, Stamford Dental Arts offers free Invisalign and implants consultations. Therefore, do not wait any longer and contact us at (203) 504–8745.
Stamford Dental Arts
44 Strawberry Hill Ave, Suite 1,
Stamford, CT 06902
(203) 504–8745
Web Address:
https://www.stamforddentalarts.com/
Our location on the map:
https://goo.gl/maps/Y2YfWteR7rjhUzUT6
https://plus.codes/87H83F68+PM Stamford
Nearby Locations:
Ridgeway | Strawberry Hill | Glenbrook | East Side Of Stamford | Downtown
06905 | 06902 | 06906 | 06901, 06905
Working Hours:
Monday: 2:00 pm — 8:00 pm
Tuesday: 8:00 am — 4:00 pm
Wednesday: 8:00 am — 4:00 pm
Thursday: 8:00 am — 8:00 pm
Friday: 8:00 am — 4:00 pm
Saturday: CLOSED
Sunday: CLOSED
Payment: cash, check, credit cards.
Directory:
Tags:
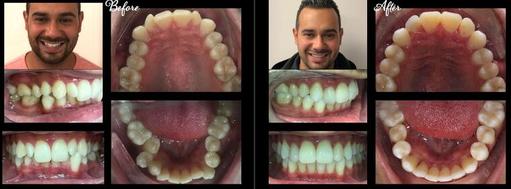
An even, appealing smile has never been easier to get at a dental center, not far away in Stamford, Connecticut. Achieve a youthful look with clear plastic trays, called aligners that do the work of metal braces without the shine. Combined with state-of-the-art, sophisticated 3D software employed by your Stamford dentists, this technology delivers straight smiles to most adults and mature teens.
The benefits of Invisalign are due to the medical-grade plastic that you can even remove the trays for short periods of time to eat, drink, brush, and floss.
You can enjoy your favorite foods with Invisalign. Go ahead, eat that apple. There is no intrusion of brackets and wires. The smooth surfaces of the plastic provide your cheeks and tongue a comfort level unlike any other form of orthodontics.
Gentle Straightening for Crooked Teeth
Invisalign pain is nearly non-existent. The slight discomfort you initially feel is due to the mild pressure on your teeth as they move. For the best and fastest results, Invisalign candidates must be willing to wear the aligners as much as possible, even approaching 24 hours a day.
Read more: https://www.stamforddentalarts.com/invisalign/
Stamford Dental Arts
44 Strawberry Hill Ave, Suite 1,
Stamford, CT 06902
(203) 504–8745
Web Address:
https://www.stamforddentalarts.com/
Our location on the map:
https://goo.gl/maps/Y2YfWteR7rjhUzUT6
https://plus.codes/87H83F68+PM Stamford
Nearby Locations:
Ridgeway | Strawberry Hill | Glenbrook | East Side Of Stamford | Downtown
06905 | 06902 | 06906 | 06901, 06905
Working Hours:
Monday: 2:00 pm — 8:00 pm
Tuesday: 8:00 am — 4:00 pm
Wednesday: 8:00 am — 4:00 pm
Thursday: 8:00 am — 8:00 pm
Friday: 8:00 am — 4:00 pm
Saturday: CLOSED
Sunday: CLOSED
Payment: cash, check, credit cards.
Directory:
Tags:
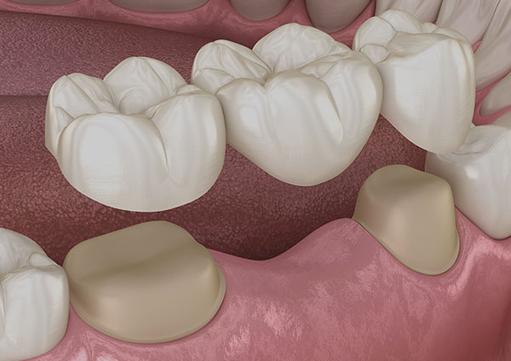
Types of Dental Bridges
A bridge can be either fixed or removable. Fixed bridges are the most common, but they necessitate crowns on both sides. These crowns are hollow, and the bridge attachment is permanently cemented into the crowns by your dentist. The artificial teeth are implanted between the crowns. A removable bridge is called a denture and is anchored by metal clasps.
Maryland bonded bridges are primarily used to replace missing front teeth. Metal wings on these resin-bonded bridges attach to your existing teeth. Traditional fixed bridges are the most common and have a lifespan of up to 15 years or more. However, when attached to dental implants, a permanent bridge can last for more than 20 years.
Read more: https://www.stamforddentalarts.com/dental-bridge/
Stamford Dental Arts
44 Strawberry Hill Ave, Suite 1,
Stamford, CT 06902
(203) 504–8745
Web Address:
https://www.stamforddentalarts.com/
Our location on the map:
https://goo.gl/maps/Y2YfWteR7rjhUzUT6
https://plus.codes/87H83F68+PM Stamford
Nearby Locations:
Ridgeway | Strawberry Hill | Glenbrook | East Side Of Stamford | Downtown
06905 | 06902 | 06906 | 06901, 06905
Working Hours:
Monday: 2:00 pm — 8:00 pm
Tuesday: 8:00 am — 4:00 pm
Wednesday: 8:00 am — 4:00 pm
Thursday: 8:00 am — 8:00 pm
Friday: 8:00 am — 4:00 pm
Saturday: CLOSED
Sunday: CLOSED
Payment: cash, check, credit cards.
Directory:
Tags:

The information provided on this site is intended to educate the reader about certain medical conditions and certain possible treatment. It is not a substitute for examination, diagnosis, and medical care provided by a licensed and qualified health care professional. If you believe you, or someone you know suffers from the conditions described herein, please see your health care provider immediately. Do not attempt to treat yourself or anyone else without proper medical supervision.
Bacterial vaginosis (BV) is the name of a condition in women where the normal balance of bacteria in the vagina is disrupted and replaced by an overgrowth of certain bacteria. It is sometimes accompanied by discharge, odor, pain, itching, or burning.
How Common Is Bacterial Vaginosis?
Bacterial vaginosis (BV) is the most common vaginal infection in women of childbearing age. In the United States, BV is common in pregnant women.
Professional Gynecological Services
3047 Brighton 6th Street #1,
Brooklyn, NY 11235
(718) 743–7877
Web Address: https://www.thewomenschoice.com/
Manhattan Beach Office: https://www.thewomenschoice.com/brighton-beach-gynecologist-nyc/
Our location on the map: https://goo.gl/maps/v5NnmgrcGbz3bGLP7
https://plus.codes/87G8H2HQ+C9 Brooklyn
Nearby Locations:
Manhattan Beach:
Brighton Beach | Manhattan Beach | Coney Island | Gravesend | Sheepshead Bay
11235 | 11224 | 11223
Working Hours:
Mon — Fri: 8:00 AM — 6:00 PM
Sat: 9:00 AM — 12:00 PM
Sun: Closed
Payment: cash, check, credit cards.
Directory:
Tags:
The information provided on this site is intended to educate the reader about certain medical conditions and certain possible treatment. It is not a substitute for examination, diagnosis, and medical care provided by a licensed and qualified health care professional. If you believe you, or someone you know suffers from the conditions described herein, please see your health care provider immediately. Do not attempt to treat yourself or anyone else without proper medical supervision.
What Is Trichomoniasis?
Trichomoniasis is a common sexually transmitted disease (STD) that affects both women and men, although symptoms are more common in women.
How Common Is Trichomoniasis?
Trichomoniasis is the most common curable STD in young, sexually active women. An estimated 7.4 million new cases occur each year in women and men.
Professional Gynecological Services
3047 Brighton 6th Street #1,
Brooklyn, NY 11235
(718) 743–7877
Web Address: https://www.thewomenschoice.com
Manhattan Beach Office: https://www.thewomenschoice.com/brighton-beach-gynecologist-nyc/
Our location on the map: https://goo.gl/maps/v5NnmgrcGbz3bGLP7
https://plus.codes/87G8H2HQ+C9 Brooklyn
Nearby Locations:
Manhattan Beach:
Brighton Beach | Manhattan Beach | Coney Island | Gravesend | Sheepshead Bay
11235 | 11224 | 11223
Working Hours:
Mon — Fri: 8:00 AM — 6:00 PM
Sat: 9:00 AM — 12:00 PM
Sun: Closed
Payment: cash, check, credit cards.
Directory:
Tags:

Professional Gynecological Services, former Manhattan Beach OGBYN, comprised of a group of physician assistants and doctors board-certified in obstetrics and gynecology, providing highly comprehensive care in a safe, compassionate, and friendly environment. We welcome you to our Brighton Beach office for convenient and expert OBGYN care. If you’re a first-time patient, make an appointment today and receive 5–10% off.
Professional Gynecological Services
3047 Brighton 6th Street #1,
Brooklyn, NY 11235
(718) 743–7877
Web Address: https://www.thewomenschoice.com
Manhattan Beach Office: https://www.thewomenschoice.com/brighton-beach-gynecologist-nyc/
Our location on the map: https://goo.gl/maps/v5NnmgrcGbz3bGLP7
https://plus.codes/87G8H2HQ+C9 Brooklyn
Nearby Locations:
Manhattan Beach:
Brighton Beach | Manhattan Beach | Coney Island | Gravesend | Sheepshead Bay
11235 | 11224 | 11223
Working Hours:
Mon — Fri: 8:00 AM — 6:00 PM
Sat: 9:00 AM — 12:00 PM
Sun: Closed
Payment: cash, check, credit cards.
Directory:
Tags:
A molar pregnancy is an abnormal pregnancy, which results from abnormal fertilization. The molar pregnancy itself is not dangerous, but if left untreated inside of the uterus it will lead to a uterine cancer, called choriocarcinoma. This means that molar pregnancy is also a precancerous condition. A molar pregnancy is also known as hydatiform mole.
As mentioned before a hydatiform mole results from an abnormal fertilization. During this abnormal fertilization, the tissue that normally forms the fetus and the placenta, in molar pregnancy will form an abnormal cystic mass. The cysts have a characteristic form of grapes.
There are two types of molar pregnancy:
– complete
– incomplete
The Difference Between a Complete and Incomplete Mole
Both complete and incomplete mole will result from an abnormal fertilization.
An complete one results from the fertilization of two spermatozoids and an egg which has no genetic material, while an incomplete mole results from the fertilization of two spermatozoids and a normal egg. The embryo created in this case will have 69 chromosomes instead of 46 chromosomes that a normal embryo has.
A complete mole only consists of a cystic mass with a characteristic form of a grape, while an incomplete mole has also fetal parts together with an abnormal cystic mass.
Both complete and incomplete mole will not lead to creation of a normal embryo and development of a normal fetus. In an incomplete molar pregnancy the embryo will begin to develop but it is malformed and can’t survive.
Professional Gynecological Services
14 Dekalb Ave, 2nd Floor,
Brooklyn, NY 11201
(718) 875–4848
Web Address: https://www.thewomenschoice.com
Our location on the map: https://goo.gl/maps/FVeCkjLkTZNiu5c16
https://plus.codes/87G8M2Q9+W3
Brooklyn
Nearby Locations:
Brooklyn:
Downtown Brooklyn | Clinton Hill | Prospect Heights | Gowanus | Cobble Hill
11201| 11238 | 11217
Working Hours:
Monday: 9 AM — 5:45 PM
Tuesday: 9 AM — 4:45 PM
Wednesday: 9 AM — 5:45 PM
Thursday: 9 AM — 4:45 PM
Friday: 9 AM — 4:45 PM
Saturday: 9 AM — 11:30AM
Sunday: Closed
Payment: cash, check, credit cards.
Directory:
Tags:
At Professional Gynecological Services we believe that patients may have a wide array of questions prior to having a termination of pregnancy. As medical providers our job is to facilitate answers to all those questions.
The following information has been designed to answer the most common questions that you may have. We invite you to read the information below carefully and want you to feel free to speak live with any of our trained medical assistants by calling 718.875.4848.
Abortion Facts
Most terminations of pregnancies are performed during the first trimester of the pregnancy. Therefore, the procedure becomes one of the safest outpatient procedures in the United States. It is eight times safer than a vaginal delivery. At Professional Gynecological Services, our complication rates are very low because of our qualified physicians and staff as well as the cleanliness and sterility throughout our office.
Each year, almost half of all pregnancies among American women are unintended. About half of these unplanned pregnancies, 1.3 million each year, are ended by abortion.
There are many myths and misconceptions about who gets abortions, and why. The fact is that the women who have abortions come from all racial, ethnic, socioeconomic and religious backgrounds.
Read more: https://www.thewomenschoice.com/surgical-abortion/
Professional Gynecological Services
14 Dekalb Ave, 2nd Floor,
Brooklyn, NY 11201
(718) 875–4848
Web Address: https://www.thewomenschoice.com
Our location on the map: https://goo.gl/maps/FVeCkjLkTZNiu5c16
https://plus.codes/87G8M2Q9+W3 Brooklyn
Nearby Locations:
Brooklyn:
Downtown Brooklyn | Clinton Hill | Prospect Heights | Gowanus | Cobble Hill
11201| 11238 | 11217
Working Hours:
Monday: 9 AM — 5:45 PM
Tuesday: 9 AM — 4:45 PM
Wednesday: 9 AM — 5:45 PM
Thursday: 9 AM — 4:45 PM
Friday: 9 AM — 4:45 PM
Saturday: 9 AM — 11:30AM
Sunday: Closed
Payment: cash, check, credit cards.







